Improved medical trial experience for patients and sponsors
Company profile
Industry
Healthcare Technology
Niche
Clinical Trial Software
Company Size
200+
HQ
Philadelphia, USA
Services provided:
A comprehensive clinical trial technology platform comprised of several sub-products and features, serving pharmaceutical companies, research organizations, and patients. The product suite includes electronic Clinical Outcome Assessment (eCOA) tools, electronic consent (eConsent) systems, and patient companion apps designed to streamline clinical trials while prioritizing participant experience and data quality.


Challenge
Clinical trial participants face complex questionnaires and consent processes that can be overwhelming and confusing. The platform needed to balance rigorous regulatory requirements with accessible, user-friendly interfaces for diverse patient populations with varying levels of digital literacy and health conditions.
Complex and unintuitive eCOA questionnaires
That were difficult for patients to understand and complete accurately, especially for the elderly or those with limited digital literacy.
Increasingly demanding compliance
Ment there was less room of errors and more stringent requirements to adhere to, complicating design decisions.
Multi-platform consistency
Required to reach a wider patient demographic, necessitating a unified design language across web and mobile. But also to reduce costs associated with logistics and training.
Diverse patient populations
Ment hat designs needed to accommodate a wide range of ages, cultural backgrounds, and health conditions, requiring adaptable and inclusive design solutions.
Solution
eCOA Questionnaires
To help expand the availability of questionnaires to new potential studies and clients, I helped redesign several complex questionnaire types, ensuring clarity, ease of use, and compliance with regulatory standards.
In-house study design platform
Helped and redesigned an internal study design platform that allows consultants to be quicker in capturing study requirements and configuring them in the system, reducing time to launch and reducing costs.
Patient Companion App
Augmented the app to also support web and tablet modalities, ensuring a seamless experience across devices. Prioritized accessibility features to accommodate diverse patient needs.
ADA compliance
Being part of a highly regulated industry with high standards for accessibility, ensured all designs met or exceeded ADA compliance guidelines to accommodate users with disabilities.
Internal ticketing system
Helped design and implement a internal change request ticketing system aimed to provide site staff with an easy way to cater to patient needs of special data changes.
Artifacts
Prioritization and roadmap

Alignment and kickoff workshop performed at the beginning of the project to ensure clarity and a better understanding of the common goals. Obscured for compliances
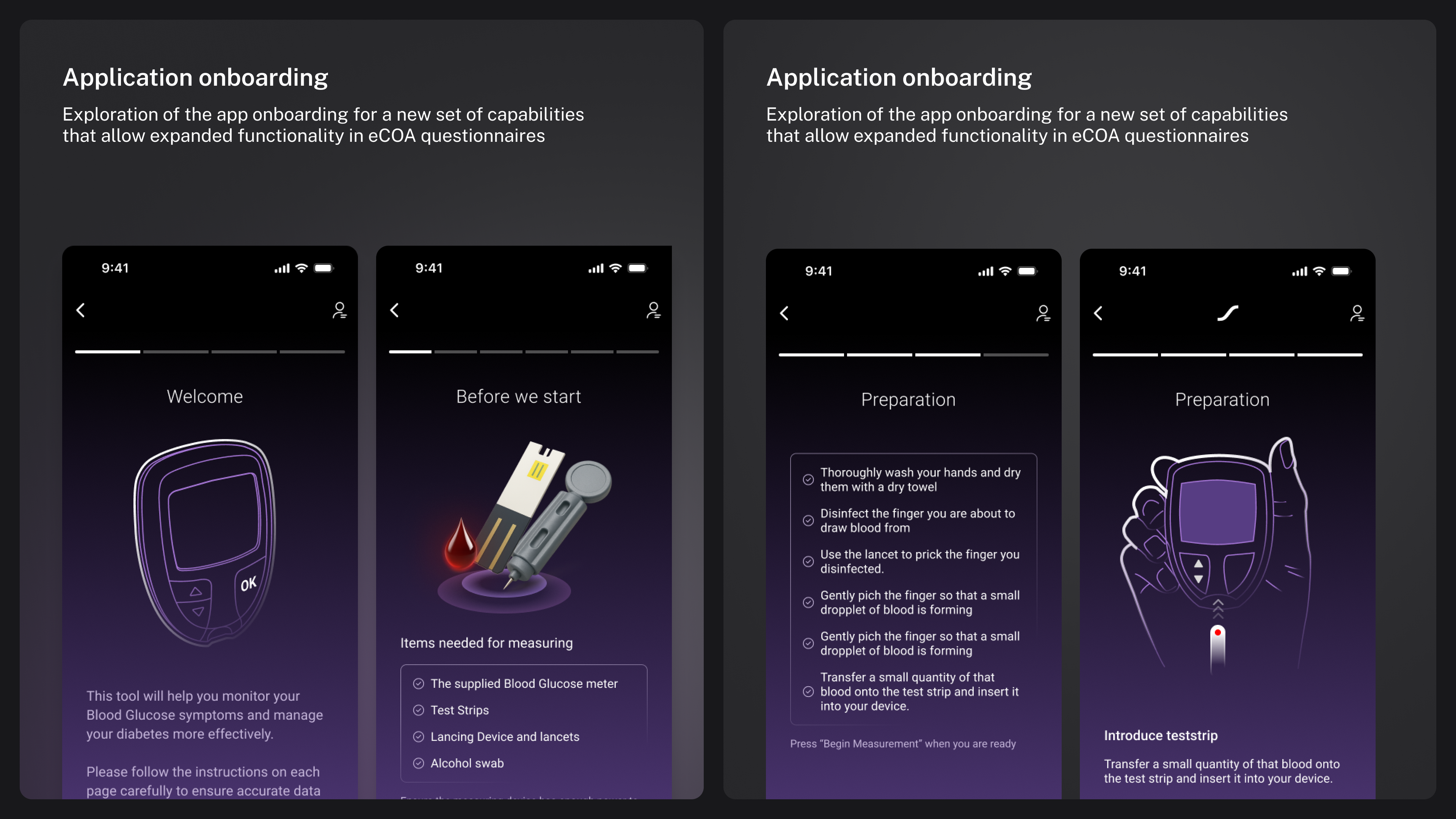
Mobile app special onboarding
Other artifacts
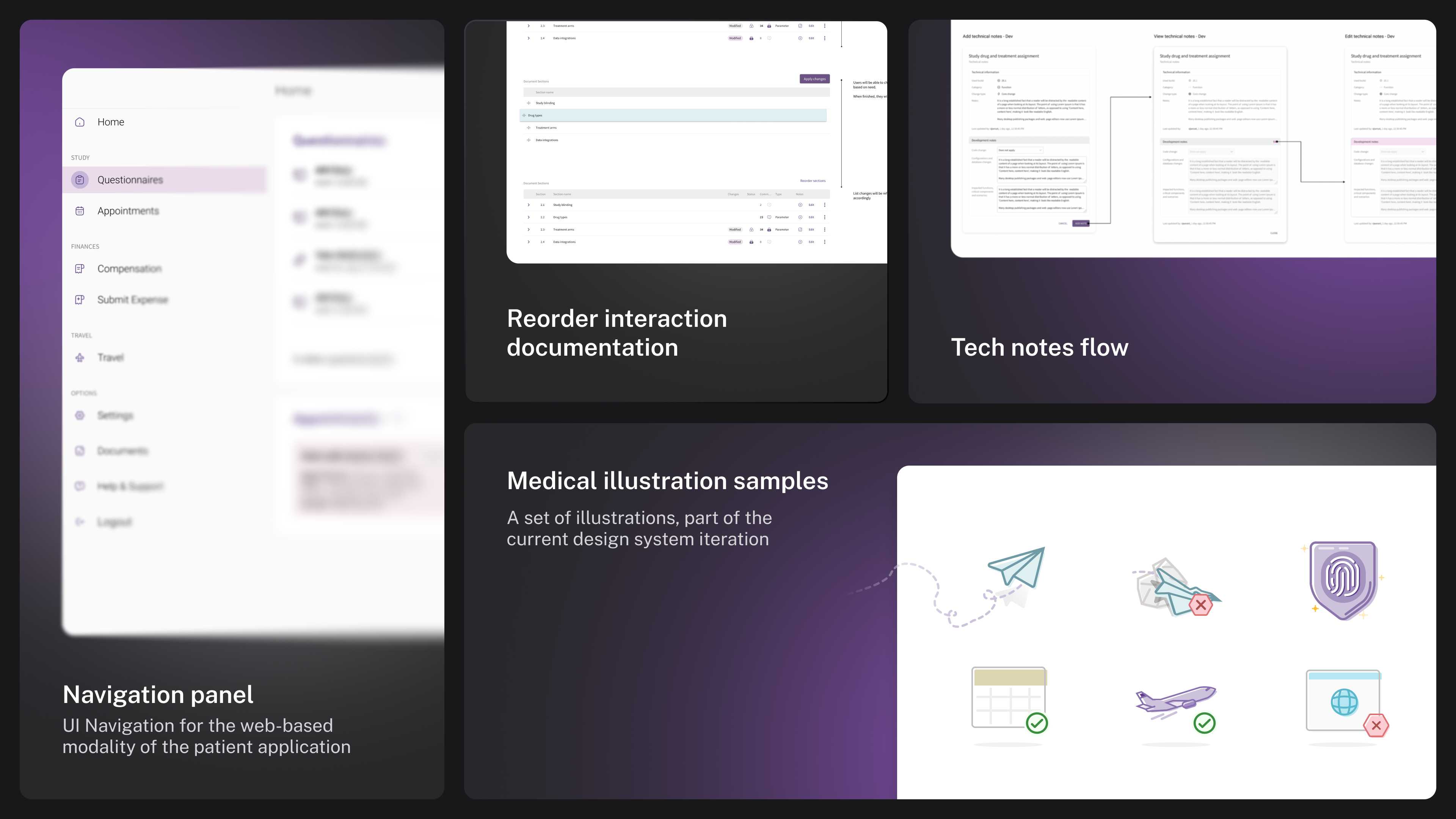
Visual assets
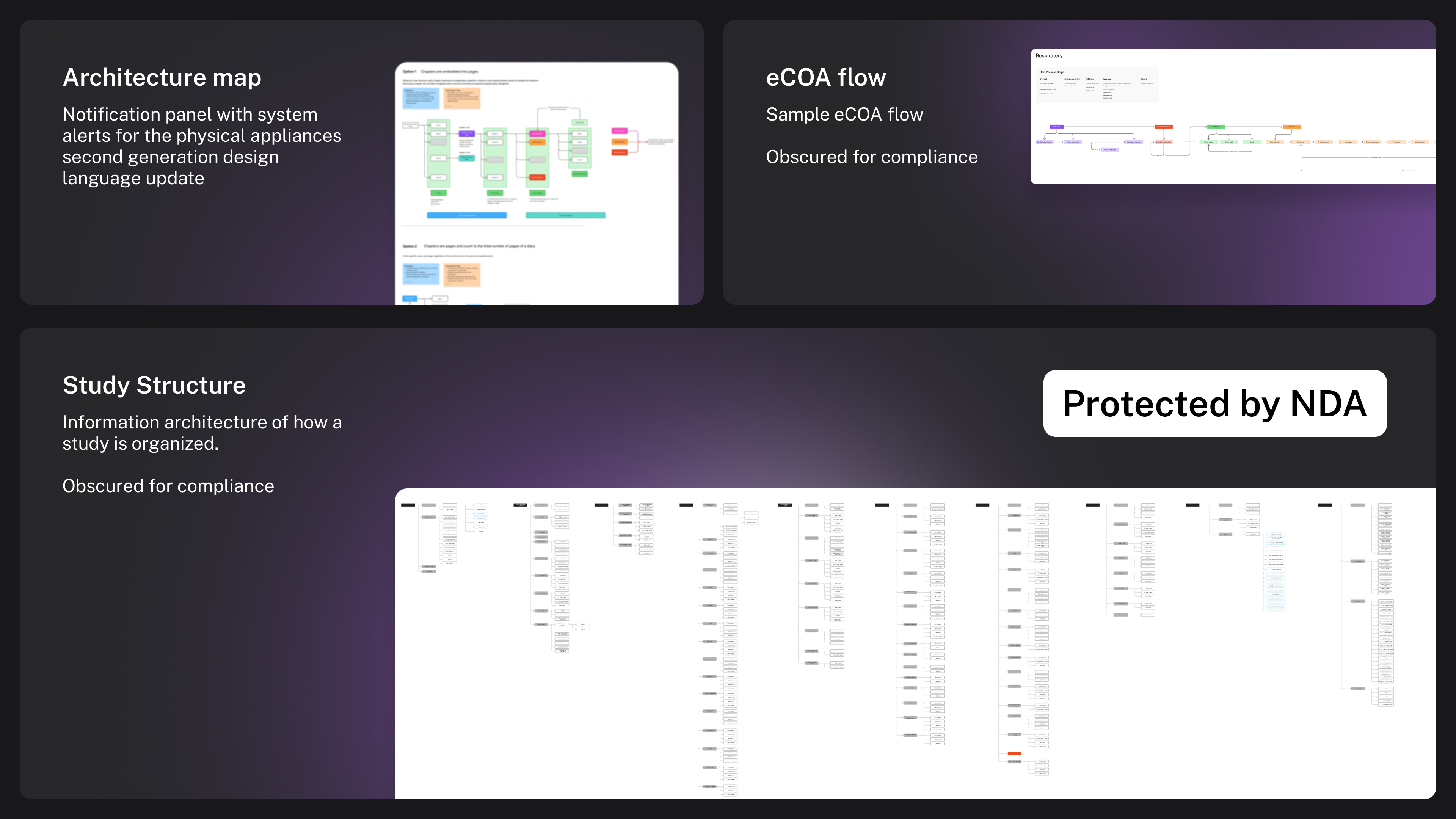
Information architecture and architecture maps
A dependency mapping concept to be used for a flow modelling functionality. Obscured for compliance.
User Flows
A complex section-based UI that is used to edit study specifications.
Results
Increased logos
In client portfolio
Signficantly increased by catering to more types of eCOA type studies that were previously inaccessible
10% less iteration
Consistent design patterns
Through the use of a internal, consistent design system, that continously brings products closer together, product recognition increased and iteration decreased.
20% increase
In revenue
To quote, "best quarter to date" for the company in terms of eCOA sales.